Born from a Surgical
"Hack" That Failed.
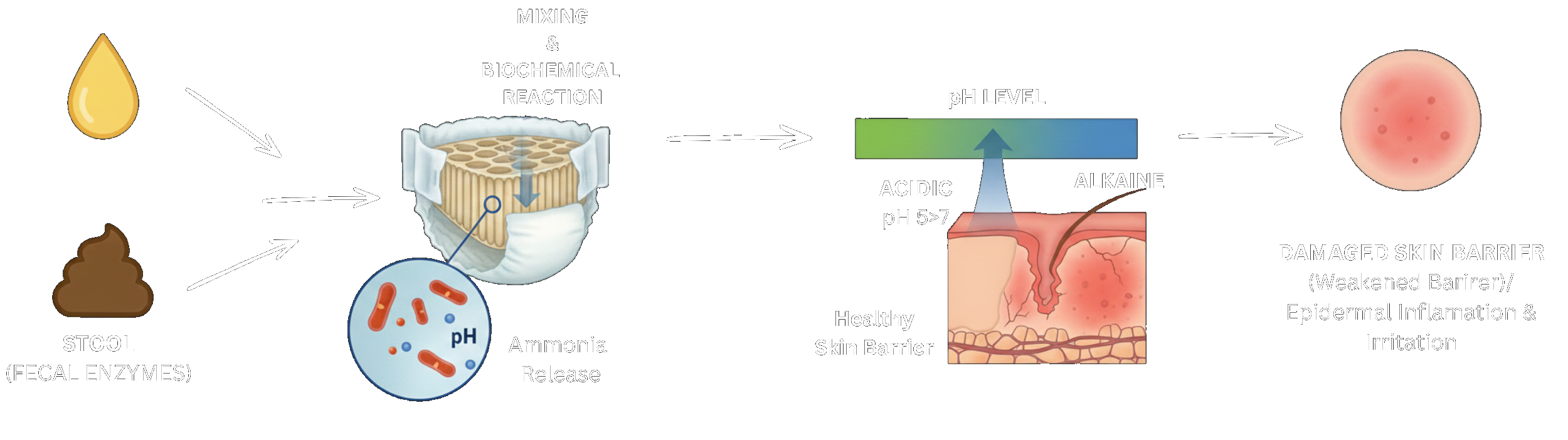
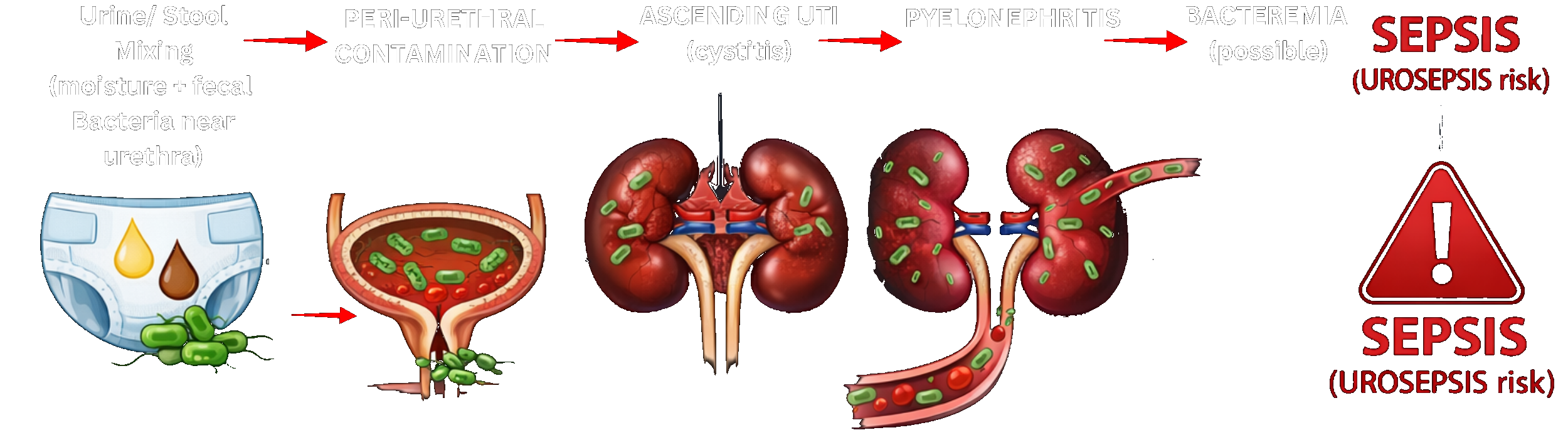
DiviDiaper Inc. started with two parents, one child, and a "standard of care" that left him completely exposed.
"What about a Divider?! What if we could separate the urine from the stool and keep it from reaching the surgical area?"— Tivon Jeffers, Co-Founder & CEO

"Just a better diaper that makes good sense — and should be used by all needing diapers."— Dr. Dix Poppas, Chief of Pediatric Urology, NewYork-Presbyterian / Weill Cornell